Stroke Article – Introduction

Martin Punter, PhD, MBChB, FRCP, FRACP, is a Consultant Neurologist at Wellington Regional Hospital, and Clinical Senior Lecturer at University of Otago Wellington, New Zealand. Martin practises general neurology as well as working in the Capital and Coast District Health Board’s stroke service for Wellington and the Central Region Telestroke Network. Martin has clinical and research interests in stroke medicine, particularly venous stroke and coordinates the 4th Year Medical Student Neurology Module in Wellington. Martin is a member of the Association of British Neurologists, the Stroke Society of Australasia, and a member of the Overseas Trained Physicians Committee for the Royal Australasian College of Physicians.
Intracerebral haemorrhage (ICH) is a particularly devastating form of stroke, affecting over 200,0000 adults around the world each year, and with higher morbidity and mortality than ischaemic stroke. Despite the huge advances in the management of ischaemic stroke, in particular hyperacute reperfusion therapies, effective treatments have historically been limited in ICH. An upcoming series of articles will explore some of the advances in treatment of ICH over the next few editions beginning with articles on the acute management of ICH.
We will kick off the series from Manchester with Alastair Paterson and Adrian Parry-Jones highlighting the latest evidence on the medical management of patients presenting with ICH in the hyper-acute phase and the need for often basic but timely intervention. Surgical management of ICH remains uncertain despite research addressing technique, technology, and timing and intervention is typically decided on a case by case basis. Chris Ovendon, Tim Kleinig and Amal Abou-Hamden from Adelaide will be presenting an update on acute ICH addressing current research on surgical techniques.
I encourage you to read these articles highlighting the latest evidence ranging from a more intensive approach to basic care to advances in surgical techniques for patients suffering from this common and devastating neurological disorder
Hyperacute Medical Management of Intracerebral Haemorrhage
Abstract
Intracerebral haemorrhage (ICH) is caused by spontaneous, non-traumatic haemorrhage within the brain parenchyma. ICH has poor outcomes, with a 30-40% 1-month case fatality and most survivors remaining dependent. Current management of ICH is aimed at providing appropriate supportive care and reducing the risk of haematoma expansion, which affects up to 20-30% of patients in the first 24 hours. Rapid and intensive blood pressure lowering to a pre-specified target and reversal of anticoagulants in the 20% of patients who are taking them may reduce the risk of expansion and improve outcome.
Intracerebral haemorrhage (ICH) is caused by spontaneous, non-traumatic haemorrhage into the brain parenchyma and globally accounts for 27.9% of incident strokes, 44% of 6.55 million stroke deaths and over half of the disability burden caused by stroke [1]. Little change in case-fatality rates within the first 48 hours of ICH onset have been seen [2]. Current treatment for ICH seeks to reduce the risk of haematoma expansion (HE), an early complication affecting up to 20-30% of ICH patients in the first 24 hours which is associated with increased morbidity and mortality.
Time since symptom onset, haematoma volume on baseline imaging and the use of anticoagulant or antiplatelet drugs at onset are all associated with a higher risk of HE. Intensive blood pressure (BP) lowering reduces haematoma expansion and along with rapid, early delivery of anticoagulant reversal, these interventions form the mainstay of acute ICH medical treatment. The antifibrinolytic agent tranexamic acid and the haemostatic drug recombinant factor VIIa (rFVIIa) reduce haematoma expansion, but have not proven overall clinical benefit in phase III clinical trials to date [3–5].
Provision of appropriate supportive care and referral of patients for neurosurgery may also be of benefit, the latter covered in another article in this series.
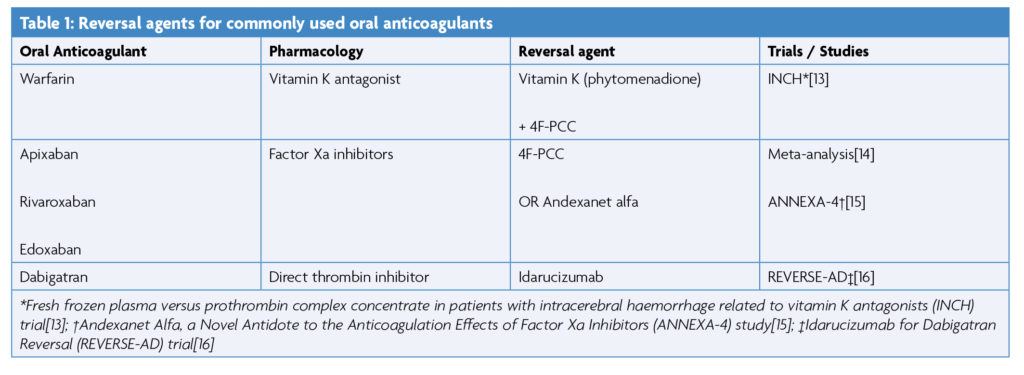
Reversing Anticoagulant Medication
Several trials have proven non-inferiority of direct oral anticoagulants (DOACs) over warfarin, a vitamin K antagonist (VKA), for stroke prevention in atrial fibrillation [3,6–8]. Lower rates of major haemorrhage and ICH are seen in patients prescribed DOACs compared with VKAs.[9] The treatment of serious bleeding associated with DOACs and VKAs is by administration of either: 4 factor prothrombin complex concentrate (4F-PCC), vitamin K, idarucizumab, or andexanet alfa, depending on which anticoagulant has been used (table 1) [10]. Correction of coagulopathy by reversal of anticoagulant medication has been linked to a reduction in HE [11]. Failure to reverse an elevated INR within 2 hours of admission is associated with an increase in morbidity and mortality [12].
VKA Reversal
VKA antagonists lead to the synthesis of ineffective clotting factors FII, FVII, FIX and FX, and anticoagulant proteins C and S. Reversal of VKAs therefore requires intravenous replacement of these factors by administration of 4F-PCC, and vitamin K (phytomenadione) administration [17]. The INCH trial found 4F-PCC to be superior to fresh frozen plasma (FFP) in normalising INR in patients with ICH [13]. Factors in 4F-PCC are 8-16 times more concentrated than in normal blood plasma and FFP, reducing the infusion volume required [18]. The use of 4F-PCC is associated with a reduced risk of 30-day mortality compared to no treatment [19].
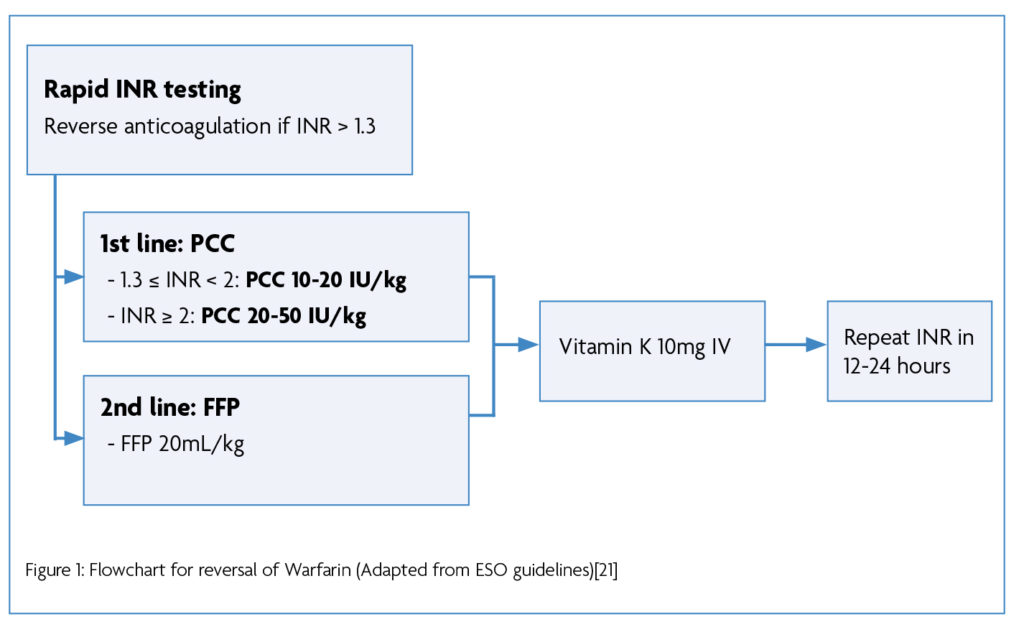
Most national and international guidance recommends vitamin K and 4F-PCC. For example, ESO guidance (2019) recommends reversal of anticoagulation if the INR >1.3. Varying doses of 4F-PCC are recommended depending on the INR, with FPP indicated as second-line. Vitamin K (10mg IV) is always indicated, and repeat INR is advised at 12-24 hours [20]. (Figure 1)

Factor Xa Inhibitor Reversal
Apixaban, rivaroxaban and edoxaban are factor Xa (FXa) inhibitors in common use. FXa inhibition prevents the FXa mediated conversion of prothrombin (II) to thrombin (IIa) [22]. 4F-PCC may be useful in replenishing FXa levels, but it does not prevent the pharmacological action of FXa inhibitors.
Andexanet alfa (AA) is a recombinant form of human factor Xa, modified to reduce its prothrombotic activity whilst retaining its Xa inhibitor binding affinity [15]. It therefore acts as a decoy receptor, binding to Xa inhibitors and preserving the function of factor Xa [23]. Although AA shows haemostatic efficacy in 90.9% of ICH patients taking apixaban and rivaroxaban [22,23], the cost of treatment stands at around £15,000 per patient [25]. NICE only recommends AA for reversal of anticoagulation in cases of life-threatening or uncontrolled gastrointestinal bleeding. NICE does not recommend the use of AA in ICH except for the purposes of research [26], as the ANNEXA-4 study under which market authorisation was applied for was a single-arm trial with high risk of bias, and was not deemed to provide sufficient evidence of cost-effectiveness [15].
The anticipated publication of the ANNEXA-I randomised controlled trial in 2025 is hoped to bring greater clarity.[27] Where AA is not available, or for factor Xa inhibitors other than apixaban and rivaroxaban, ESO and AHA guidelines recommend the use of 4F-PCC (37.5-50 IU/kg) [20,28].
Direct Thrombin Inhibitor Reversal
Dabigatran is a direct thrombin inhibitor. Reversal of this agent is with idarucizumab (Praxbind), a monoclonal antibody fragment. Both ESO and AHA guidelines recommend the use of idarucizumab for dabigatran therapy reversal (2 x 2.5g IV) as it was found to completely reverse the anticoagulant action of dabigatran within minutes of administration in the RE-VERSE AD trial [16].
Blood Pressure Lowering
The implementation of intensive BP lowering appears to reduce HE when compared with guideline treatment, although this reduction is not associated with any improvement in functional recovery at 3-6 months [29]. A number of trials have examined the impact of systolic blood pressure reduction on death and disability, using a range of approaches and agents [30–35].
Two key trials, INTERACT-2[30] and ATACH-2 [36], produced conflicting results, reporting a marginal reduction versus no reduction in death and major disability respectively. Participants in ATACH-2 had a larger SBP reduction, which may have increased the risk of adverse events in this trial. In a pooled analysis of the two trials, Wang et al. identified a J-shaped curve between SBP reduction and odds of a poor outcome at 90 days. This demonstrates that while improved outcomes were seen in SBP reduction up to 32mmHg, this association began to reverse at 46mmHg, and a SBP reduction of >72mmHg was associated with a poor outcome.
The risk of Acute Kidney Injury (AKI) more than doubled where SBP reduction was >90mmHg, compared to <90mmHg. In patients with CKD, this risk almost quadrupled [37]. Larger decreases in eGFR were associated with AKI and higher modified Rankin Scale (mRS) scores [38]. Most guidelines recommend the intervention tested in INTERACT-2 or something similar – that is to reduce the SBP to < 140 mmHg within 1 hour for patients presenting within 6 hours of onset and with an SBP > 150 mmHg [39–41]. Due to a lack of evidence, optimal management for patients beyond 6 hours and for those with unknown time of onset is not clear thus guidelines vary in their recommendations.
Supportive Care
Fast, evidence-based, proactive and targeted control of haemodynamics and blood pressure whilst considering neurosurgical input has been linked to improved outcomes. The delivery of an ‘ABC’ care bundle (Anticoagulant reversal, BP lowering, Care pathway for referral to neurosurgery) at one stroke centre in the UK was associated with a reduced needle-to-target time for BP reduction and a reduced 30-day case-fatality rate [42]. Lower rates of early do not resuscitate (DNR) orders and improved access to critical care were also observed in this study [43].
Meta-analysis data supports the management of ICH in specialist stroke care settings: a greater reduction in mortality was seen in patients with haemorrhagic stroke than ischaemic stroke when both groups were treated on stroke units [44].
Conclusion
The medical management of ICH is focused on reducing the risk of HE through reversal of anticoagulants and intensive BP lowering. Improving the management of ICH is not solely limited to newer and more costly medications. Improving the timeliness and intensity of care in relation to BP management and anticoagulant reversal whilst assigning patients to the appropriate care pathways shows promise for future hyperacute care in ICH.
References
- Feigin VL, Stark BA, Johnson CO, Roth GA, Bisignano C, Abady GG, et al. Global, regional, and national burden of stroke and its risk factors, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Neurol. 2021 Oct 1;20(10):795-820.
- Béjot Y, Grelat M, Delpont B, Durier J, Rouaud O, Osseby GV, et al. Temporal trends in early case-fatality rates in patients with intracerebral hemorrhage. Neurology. 2017 Mar 7;88(10):985-90. https://doi.org/10.1212/WNL.0000000000003681
- Connolly SJ, Ezekowitz MD, Yusuf S, Eikelboom J, Oldgren J, Parekh A, et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med. 2009 Sep 17;361(12):1139-51. https://doi.org/10.1056/NEJMoa0905561
- Sprigg N, Flaherty K, Appleton JP, Salman RAS, Bereczki D, Beridze M, et al. Tranexamic acid for hyperacute primary IntraCerebral Haemorrhage (TICH-2): an international randomised, placebo-controlled, phase 3 superiority trial. The Lancet. 2018 May 26;391(10135):2107-15. https://doi.org/10.1016/S0140-6736(18)31033-X
- Mayer SA, Brun NC, Begtrup K, Broderick J, Davis S, Diringer MN, et al. Efficacy and Safety of Recombinant Activated Factor VII for Acute Intracerebral Hemorrhage. N Engl J Med. 2008 May 15;358(20):2127-37. https://doi.org/10.1056/NEJMoa0707534
- Patel MR, Mahaffey KW, Garg J, Pan G, Singer DE, Hacke W, et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med. 2011 Sep 8;365(10):883-91. https://doi.org/10.1056/NEJMoa1009638
- Granger CB, Alexander JH, McMurray JJV, Lopes RD, Hylek EM, Hanna M, et al. Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med. 2011 Sep 15;365(11):981-92. https://doi.org/10.1056/NEJMoa1107039
- Giugliano RP, Ruff CT, Braunwald E, Murphy SA, Wiviott SD, Halperin JL, et al. Edoxaban versus warfarin in patients with atrial fibrillation. N Engl J Med. 2013 Nov 28;369(22):2093-104. https://doi.org/10.1056/NEJMoa1310907
- Kjerpeseth LJ, Ellekjær H, Selmer R, Ariansen I, Furu K, Skovlund E. Trends in use of warfarin and direct oral anticoagulants in atrial fibrillation in Norway, 2010 to 2015. Eur J Clin Pharmacol. 2017 Nov 1;73(11):1417-25. https://doi.org/10.1007/s00228-017-2296-1
- Kuramatsu JB, Sembill JA, Huttner HB. Reversal of oral anticoagulation in patients with acute intracerebral hemorrhage. Crit Care. 2019 Jun 6;23(1):206. https://doi.org/10.1186/s13054-019-2492-8
- Sweidan AJ, Singh NK, Conovaloff JL, Bower M, Groysman LI, Shafie M, et al. Coagulopathy reversal in intracerebral haemorrhage. Stroke Vasc Neurol [Internet]. 2020 Mar 1 [cited 2022 Apr 13];5(1). Available from: https://svn.bmj.com/content/5/1/29 https://doi.org/10.1136/svn-2019-000274
- Bower MM, Sweidan AJ, Shafie M, Atallah S, Groysman LI, Yu W. Contemporary Reversal of Oral Anticoagulation in Intracerebral Hemorrhage. Stroke. 2019 Feb;50(2):529-36. https://doi.org/10.1161/STROKEAHA.118.023840
- Steiner T, Poli S, Griebe M, Hüsing J, Hajda J, Freiberger A, et al. Fresh frozen plasma versus prothrombin complex concentrate in patients with intracranial haemorrhage related to vitamin K antagonists (INCH): a randomised trial. Lancet Neurol. 2016 May 1;15(6):566-73. https://doi.org/10.1016/S1474-4422(16)00110-1
- Majeed A, Ågren A, Holmström M, Bruzelius M, Chaireti R, Odeberg J, et al. Management of rivaroxaban- or apixaban-associated major bleeding with prothrombin complex concentrates: a cohort study. Blood. 2017 Oct 12;130(15):1706-12. https://doi.org/10.1182/blood-2017-05-782060
- Connolly SJ, Crowther M, Eikelboom JW, Gibson CM, Curnutte JT, Lawrence JH, et al. Full Study Report of Andexanet Alfa for Bleeding Associated with Factor Xa Inhibitors. N Engl J Med. 2019 Apr 4;380(14):1326-35. https://doi.org/10.1056/NEJMoa1814051
- Pollack CV, Reilly PA, van Ryn J, Eikelboom JW, Glund S, Bernstein RA, et al. Idarucizumab for Dabigatran Reversal – Full Cohort Analysis. N Engl J Med. 2017 Aug 3;377(5):431-41. https://doi.org/10.1056/NEJMoa1707278
- Holt T, Taylor S, Abraham P, Mcmillian W, Harris S, Curtis J, et al. Three- versus four-factor prothrombin complex concentrate for the reversal of warfarin-induced bleeding. Int J Crit Illn Inj Sci. 2018;8(1):36-40. https://doi.org/10.4103/IJCIIS.IJCIIS_40_17
- Herzog E, Kaspereit F, Krege W, Niebl P, Schulte S, Dickneite G. Four-Factor Prothrombin Complex Concentrate (4F-PCC) Is Superior to Three-Factor Prothombin Comlex Concentrates (3F-PCC) for Reversal of Coumarin Anticoagulation. Blood. 2014 Dec 6;124(21):1472. https://doi.org/10.1182/blood.V124.21.1472.1472
- Parry-Jones AR, Di Napoli M, Goldstein JN, Schreuder FHBM, Tetri S, Tatlisumak T, et al. Reversal strategies for vitamin K antagonists in acute intracerebral hemorrhage. Ann Neurol. 2015 Jul;78(1):54-62. https://doi.org/10.1002/ana.24416
- Christensen H, Cordonnier C, Kõrv J, Lal A, Ovesen C, Purrucker JC, et al. European Stroke Organisation Guideline on Reversal of Oral Anticoagulants in Acute Intracerebral Haemorrhage. Eur Stroke J. 2019 Dec 1;4(4):294-306. https://doi.org/10.1177/2396987319849763
- Steiner T, Salman RAS, Beer R, Christensen H, Cordonnier C, Csiba L, et al. European Stroke Organisation (ESO) Guidelines for the Management of Spontaneous Intracerebral Hemorrhage. Int J Stroke. 2014 Oct 1;9(7):840-55. https://doi.org/10.1111/ijs.12309
- Harder S, Graff J. Novel oral anticoagulants: clinical pharmacology, indications and practical considerations. Eur J Clin Pharmacol. 2013 Sep 1;69(9):1617-33. https://doi.org/10.1007/s00228-013-1510-z
- Kaatz S, Bhansali H, Gibbs J, Lavender R, Mahan CE, Paje DG. Reversing factor Xa inhibitors – clinical utility of andexanet alfa. J Blood Med. 2017 Sep 13;8:141-9. https://doi.org/10.2147/JBM.S121550
- Siegal DM, Curnutte JT, Connolly SJ, Lu G, Conley PB, Wiens BL, et al. Andexanet Alfa for the Reversal of Factor Xa Inhibitor Activity. N Engl J Med. 2015 Dec 17;373(25):2413-24. https://doi.org/10.1056/NEJMoa1510991
- Information about andexanet alfa | Andexanet alfa for reversing anticoagulation from apixaban or rivaroxaban | Andexanet alfa for reversing anticoagulation from apixaban or rivaroxaban [ID1101] | Consultations | NICE [Internet]. [cited 2022 Jun 12]. Available from: https://www.nice.org.uk/consultations/1058/3/information-about-andexanet-alfa
- Recommendations | Andexanet alfa for reversing anticoagulation from apixaban or rivaroxaban | Guidance | NICE [Internet]. NICE; [cited 2022 Jun 12]. Available from: https://www.nice.org.uk/guidance/ta697/chapter/1-Recommendations
- Alexion Pharmaceuticals. A Randomized Clinical Trial of Andexanet Alfa in Acute Intracranial Hemorrhage in Patients Receiving an Oral Factor Xa Inhibitor [Internet]. clinicaltrials.gov; 2022 Jun [cited 2022 Jun 9]. Report No.: NCT03661528. Available from: https://clinicaltrials.gov/ct2/show/NCT03661528.
- Greenberg SM, Ziai WC, Cordonnier C, Dowlatshahi D, Francis B, Goldstein JN, et al. Guideline for the Management of Patients With Spontaneous Intracerebral Hemorrhage: A Guideline From the American Heart Association/American Stroke Association. Stroke. 2022. https://doi.org/10.1161/STR.0000000000000407
- Moullaali TJ, Wang X, Sandset EC, Woodhouse LJ, Law ZK, Arima H, et al. Early lowering of blood pressure after acute intracerebral haemorrhage: a systematic review and meta-analysis of individual patient data. J Neurol Neurosurg Psychiatry. 2022 Jan 1;93(1):6-13. https://doi.org/10.1136/jnnp-2022-ABN.359
- Anderson CS, Huang Y, Wang JG, Arima H, Neal B, Peng B, et al. Intensive blood pressure reduction in acute cerebral haemorrhage trial (INTERACT): a randomised pilot trial. Lancet Neurol. 2008 May;7(5):391-9. https://doi.org/10.1016/S1474-422(08)70069-
- Delcourt C, Huang Y, Wang J, Heeley E, Lindley R, Stapf C, et al. The Second (Main) Phase of an Open, Randomised, Multicentre Study to Investigate the Effectiveness of an Intensive Blood Pressure Reduction in Acute Cerebral Haemorrhage Trial (Interact2). Int J Stroke. 2010 Apr 1;5(2):110-6. https://doi.org/10.1111/j.1747-4949.2010.00415.x
- Bath PM, Scutt P, Anderson CS, Appleton JP, Berge E, Cala L, et al. Prehospital transdermal glyceryl trinitrate in patients with ultra-acute presumed stroke (RIGHT-2): an ambulance-based, randomised, sham-controlled, blinded, phase 3 trial. The Lancet. 2019 Mar 9;393(10175):1009-20.
- Efficacy of nitric oxide, with or without continuing antihypertensive treatment, for management of high blood pressure in acute stroke (ENOS): a partial-factorial randomised controlled trial. The Lancet. 2015 Feb 14;385(9968):617-28. https://doi.org/10.1016/S0140-6736(14)61121-1
- Antihypertensive Treatment of Acute Cerebral Hemorrhage (ATACH) investigators. Antihypertensive treatment of acute cerebral hemorrhage. Crit Care Med. 2010 Feb;38(2):637-48. https://doi.org/10.1097/CCM.0b013e3181b9e1a5
- Qureshi AI, Palesch YY, Barsan WG, Hanley DF, Hsu CY, Martin RL, et al. Intensive Blood-Pressure Lowering in Patients with Acute Cerebral Hemorrhage. N Engl J Med. 2016 Sep 15;375(11):1033-43.https://doi.org/10.1056/NEJMoa1603460
- Qureshi AI, Palesch YY. Antihypertensive Treatment of Acute Cerebral Hemorrhage (ATACH) II: design, methods, and rationale. Neurocrit Care. 2011 Dec;15(3):559-76. https://doi.org/10.1007/s12028-011-9538-3
- Burgess LG, Goyal N, Jones GM, Khorchid Y, Kerro A, Chapple K, et al. Evaluation of Acute Kidney Injury and Mortality After Intensive Blood Pressure Control in Patients With Intracerebral Hemorrhage. J Am Heart Assoc Cardiovasc Cerebrovasc Dis. 2018 Apr 13;7(8):e008439. https://doi.org/10.1161/JAHA.117.008439
- Zheng D, Sato S, Arima H, Heeley E, Delcourt C, Cao Y, et al. Estimated GFR and the Effect of Intensive Blood Pressure Lowering After Acute Intracerebral Hemorrhage. Am J Kidney Dis Off J Natl Kidney Found. 2016 Jul;68(1):94-102.
- Stroke and transient ischaemic attack in over 16s: diagnosis and initial management (NG128) [Internet]. National Institute for Health and Care Excellence [NICE]. NICE; 2019 [cited 2022 Apr 11]. Available from: https://www.nice.org.uk/guidance/ng128/chapter/Recommendations#blood-pressure-control-for-people-with-acute-intracerebral-haemorrhage
- Hemphill JC, Greenberg SM, Anderson CS, Becker K, Bendok BR, Cushman M, et al. Guidelines for the Management of Spontaneous Intracerebral Hemorrhage. Stroke. 2015 Jul;46(7):2032-60. https://doi.org/10.1161/STR.0000000000000069
- Sandset EC, Anderson CS, Bath PM, Christensen H, Fischer U, Gąsecki D, et al. European Stroke Organisation (ESO) guidelines on blood pressure management in acute ischaemic stroke and intracerebral haemorrhage. Eur Stroke J. 2021 Jun 1;6(2):XLVIII-LXXXIX. https://doi.org/10.1177/23969873211012133
- Parry-Jones AR, Sammut-Powell C, Paroutoglou K, Birleson E, Rowland J, Lee S, et al. An Intracerebral Hemorrhage Care Bundle Is Associated with Lower Case Fatality. Ann Neurol. 2019;86(4):495-503. https://doi.org/10.1002/ana.25546
- Parry-Jones AR, Moullaali TJ, Ziai WC. Treatment of intracerebral hemorrhage: From specific interventions to bundles of care. Int J Stroke. 2020 Dec 1;15(9):945-53. https://doi.org/10.1177/1747493020964663
- Langhorne P, Fearon P, Ronning OM, Kaste M, Palomaki H, Vemmos K, et al. Stroke unit care benefits patients with intracerebral hemorrhage: systematic review and meta-analysis. Stroke. 2013 Nov;44(11):3044-9. https://doi.org/10.1161/STROKEAHA.113.


