The upper limb and stroke
Arm weakness is common after stroke and its treatment is recognised as an area of considerable need.1 Approximately 85% of patients with stroke present with arm weakness2 and 60% of stroke survivors with poorly functioning arms at one week do not recover meaningful function by six months.3 Arm weakness is a major factor contributing to disability following stroke.4 Current treatment for arm weakness typically comprises intensive, task-specific and repetitive rehabilitative interventions or occasionally methods such as constraint induced movement therapy and robotic therapy.5 A recent meta-analysis and large-scale trials show the effects of current treatments for arm weakness to be modest.6,7 Improvement in arm function should improve quality of life for stroke survivors, reduce co-morbidities associated with loss of independence, and reduce cost to the health care system.8
Neuroplasticity and recovery
Neuroplasticity is the brain’s ability to form new neural pathways in response to injury or disease. It has been a target for the treatment of many neurological disorders including epilepsy and tinnitus. Recent studies have suggested that augmentation of neuroplasticity is required to more fully recover motor function.9 Novel techniques that drive the growth of new neural pathways related to motor function are needed; vagus nerve stimulation (VNS) may achieve this.
Vagus nerve stimulation
VNS is the delivery of small electrical impulses to the vagus nerve (Figure 1). VNS activates neurons in the basal forebrain and locus coeruleus and results in the release of acetylcholine and norepinephrine. These neurotransmitters are known to facilitate the reorganisation of cortical networks.10 VNS is already used to treat patients with medically refractory epilepsy, with studies showing a reduction in seizure frequency of 50% in 24.5 to 46.6% of patients.11,12,13 In excess of 75,000 patients with refractory epilepsy have been implanted with VNS devices.14 The concept of using VNS to restore normal neuronal activity / drive neuroplasticity is under investigation in other chronic neurological conditions.
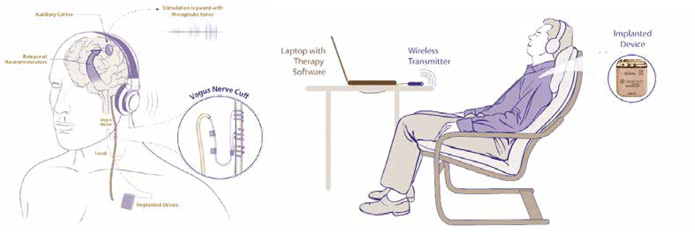
In noise induced tinnitus, cochlear trauma can lead to a disorganised auditory cortex resulting in chronic symptoms.15,16,17 The severity of tinnitus is related to the degree of map re-organisation in the auditory cortex.15,16,17 In pre-clinical studies, pairing auditory tones with brief pulses of VNS has been shown to cause re-organisation of auditory cortex maps specific to that tone.18 Further, noise-exposed rats were noted to have a significant reduction in startle response, presumably due to tinnitus, and pairing VNS with multiple tones reversed this effect.18 Thus, VNS paired with a specific stimulus may drive neuroplasticity specifically for that stimulus, thereby restoring auditory cortex architecture and reducing tinnitus. Studies suggest that VNS may help humans with tinnitus.19 Ten patients known to have unilateral or bilateral tinnitus for over a year received four weeks of VNS paired with auditory tone therapy (using MicroTransponder Inc’s Serenity© system). Subjective and objective primary outcome measures were identified in the form of the Tinnitus Handicap Inventory (THI) and the Minimum Masking Level (MML). In patients who had not been taking drugs which could interfere with VNS (muscarinic antagonists, noradrenergic reuptake inhibitors and γ-amino butyric acid agonists), a significant fall in THI of 28.17% was seen following VNS paired with auditory tones.19 Three out of five such patients had a clinically meaningful decrease in THI (44.3% decrease).19 Similar results were seen in the MML test which detects the lowest level of noise required to “drown out” the tinnitus. Results of a recently completed and larger, double blind and randomised study of VNS paired with auditory tones in tinnitus are eagerly awaited. Another study looked at the use of transcutaneous vagus nerve (t-VNS) stimulation in tinnitus. When used in combination with sound therapy t-VNS was found to modulate auditory cortical activation, resulting in reduced tinnitus and tinnitus associated distress.20
Following on from the work in tinnitus, it is hypothesised that VNS paired with upper limb rehabilitation could drive neuroplasticity specific for upper limb tasks and improve outcomes for stroke survivors. Recent animal studies have shown that VNS paired with motor rehabilitation improves forelimb function after stroke more than either rehabilitation or VNS alone.21,22,23 Pairing VNS with motor tasks induces plasticity in the motor cortex that is specific to the paired movement and is not seen with motor training alone (Figure 2).24 The timing and amount of VNS appear important to ensure optimal results are achieved; delaying VNS until after rehabilitation and significantly increasing the amount of VNS delivered results in a poorer outcome.21,23 Similar results have been seen in experimental models of haemorrhagic stroke; a 77% recovery in function was seen in the VNS plus rehabilitation group as opposed to a 29% recovery in the rehabilitation only group, which was sustained six weeks after VNS was stopped.22
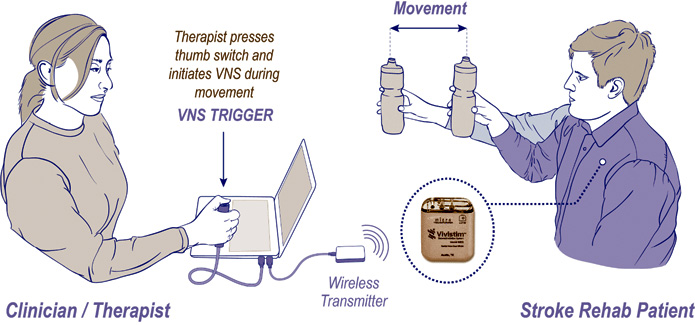
The first in-human evaluation of VNS paired with upper limb rehabilitation was recently completed using MicroTransponder Inc’s Vivistim© system. VNS treatment for stroke involves high intensity repetitive rehabilitation tasks and each movement task is paired with a 0.5 second train of VNS (0.8 mA). Each two hour long therapy session typically involves 400 such stimulations. Twenty-one patients with an ischaemic stroke and upper limb weakness were randomised to six weeks of VNS plus rehabilitation or rehabilitation alone. The per-protocol analysis included participants who attended at least 12 of the 18 therapy sessions and were not on medications that could interfere with VNS. The upper extremity Fugl-Meyer (FMA-UE) score was used to compare outcomes in the two groups, although the primary objective was to assess safety and feasibility of treatment. A clinically significant improvement was defined as a change in FMA-UE score of six or more. In the per-protocol analysis the mean change in FMA-UE score was 9.6 points in the VNS group and 3.0 points in the rehabilitation only group. Six (66·7%) achieved a clinically meaningful response on the FMA-UE score in the VNS group compared to 4 (36·4%) in the rehabilitation only group (p=0·17).25 The study confirmed that VNS paired with rehabilitation is feasible and did not raise safety concerns.25 A further randomised double blind and sham controlled study is under way.
Suitability of VNS therapy after stroke
Clearly further study is needed. VNS is already used safely and effectively in patients with medically refractory epilepsy and in the stroke stimulation paradigm, only approximately 1% of the VNS given in epilepsy is used (the device is only active during therapy). This may further reduce the risk of adverse events. Little additional set up time is required and therapists can be readily trained to deliver VNS therapy in contrast to techniques such as transcranial magnetic stimulation. One of the main disadvantages is that surgery is required, which will exclude some patients and carries small risks of infection, vocal cord palsy or anaesthetic complications. The use of t-VNS has not yet been explored after stroke but has been studied in both tinnitus and epilepsy.
Summary and further research implications
Animal studies have shown that VNS paired with rehabilitation therapy for the upper limb augments upper limb specific neuroplasticity and improves forelimb function. A small pilot study in humans has shown promising improvements in upper limb function and confirmed it is an acceptably safe and feasible treatment for further study. Further research is needed to assess efficacy as the recent clinical pilot study was small.
References
- Pollock A, St George B, Firkins L. Top ten research priorities relating to life after stroke. Lancet Neurology. 2012;11:209.
- Nakayama H, Jorgenson HS, Raaschou HO, Olsen TS. Recovery of upper extremity function in stroke patients: the Copenhagen Stroke Study. Arch Phys Med Rehabil. 1194;75:394-8.
- Wade DT, Langton-Hewer R, Wood VA, Skilbeck CE, Ismail HM. The hemiplegic arm after stroke: measurement and recovery. Journal of Neurology, Neurosurgery and Psychiatry 1983;46:521-4.
- Canning CG, Ada L, Adams R, O’Dwyer NJ. Loss of strength contributes more to physical disability after stroke than loss of dexterity. Clin Rehabil. 2004; 18:300-8.
- National Institute for Health Care and Clinical Excellence, Stroke Long term rehabilitation after stroke. CG162, London. National Institute for Health Care and Clinical Excellence. 2013.
- Langhorne P, Coupar F, Pollock A. Motor recovery after stroke: a systematic review. Lancet Neurology. 2009:8:741-54.
- Lo A, Guarino PD, Richards LG, Haselkorn JK, Wittenberg GF, Federman DG, et al. 333 2010;362:1772-83.
- Wagner TH, Lo AC, Peduzzi P, Bravata DM. An economic analysis of robot-assisted therapy for long-term upper-limb impairment after stroke. 2011;42:2630-32.
- Cramer SC. Sur M, Dobkin BH, O’Brien C, Sanger TD, Trojanowski JQ, et al. Harnessing neuroplasticity for clinical applications. 2011;134:1591-609.
- Nichols JA, Nichols AR, Smirnakis SM, Engineer NS, Kilgard MP, Atzori M. Vagus nerve stimulation modulates cortical synchrony and excitability through the activation of muscarinic receptors. 2011;189:207-14.
- Ben-Menachem E, Manon-Espaillat R, Ristanovic R. Vagus nerve stimulation for treatment of partial seizures: 1. A controlled study of effect on seizures. First International Vagus Nerve Stimulation Study Group. Epilepsia 1994;35:616-26.
- Vagus nerve Stimulation Study Group. A randomised controlled trial of chronic vagus nerve stimulation for treatment of medically intractable seizures. Neurology 1995;45:224-30.
- Uthman BM, Wilder BJ, Penry JK. Treatment of epilepsy by stimulation of the vagus nerve. Neurology 1993;43:1338-45.
- Cyberonics Inc 2013 Annual Report
- Flor H, Elbert T, Knecht S, Wienbruch C, Pantev C, Birbaumer N, Larbig W, Taub E. Phantom-limb pain as a perceptual correlate of cortical reorganization following arm amputation. 1995 Jun 8;375(6531):482-4.
- Eggermont JJ, Roberts LE. The neuroscience of tinnitus. Trends Neurosci. 2004 Nov;27(11):676-82.
- Møller AR. Tinnitus and pain. Prog Brain Res. 2007;166:47-53.
- Engineer ND, Riley JR, Seale JD, Vrana WA, Shetake JA, Sudanagunta SP, et al. Reversing pathological neural activity using targeted plasticity. 2011;470:101-04.
- De Ridder D, Vanneste S, Engineer ND, Kilgard MP. Safety and Efficacy of Vagus Nerve Stimulation Paired With Tones for the Treatment of Tinnitus: A Case Series. 2014;17:170-79.
- Lehtimäki J, Hyvärinen P, Ylikoski M, Bergholm M, Mäkelä JP, Aarnisalo A, Pirvola U, Mäkitie A, Ylikoski J. Transcutanous nerve stimulation in tinnitus: a pilot study. Acta Otolaryngol. 2013;144(4):378-82.
- Khodaparast N, Hays SA, Sloan AM, Fayyaz T, Hulsey DR, Rennaker RL 2nd, et al. Vagus nerve stimulation delivered during motor rehabilitation improves recovery in a rat model of stroke. Neurorehabil Neural Repair. 2014;28:698-706.
- Hays SA, Khodaparest N, Hulsey D, Ruiz A, Sloan AM, Rennaker RL 2nd, et al. Vagus nerve stimulation during rehabilitative training improves functional recovery after intracerebral hemorrhage. 2014;45:3097–100.
- Hays SA, Khodaparast N, Ruiz A, Sloan AM, Hulsey DR, Rennaker RL 2nd, et al. The timing and amount of vagus nerve stimulation during rehabilitative training affect post stroke recovery of forelimb strength. 2014; 25:676-82.
- Porter BA, Khodaparast N, Fayyaz T, Cheung RJ, Ahmed SS, Vrana WA, et al. Repeatedly pairing vagus nerve stimulation with a movement reorganizes primary motor cortex. Cereb Cortex. 2012;22:2365-74.
- Dawson J, Pierce D, Dixit A, Kimberley TJ, Robertson M, Tarver B, Hilmi O, McLean J, Forbes K, Kilgard MP, Rennaker RL, Cramer SC, Walters M, Engineer N. Safety, Feasibility, and Efficacy of Vagus Nerve Stimulation Paired With Upper-Limb Rehabilitation After Ischemic Stroke. 2016 Jan;47(1):143-50.

