This article has been supplied by Nestlé Health Science and they have supported the costs of its publication.
According to new research conducted by the University of Sherbrooke in Canada, in partnership with Nestlé Health Science and published in Alzheimer’s & Dementia: The Journal of the Alzheimer’s Association, regular consumption of Nestlé Health Science’s BrainXpert, a specialised ketogenic drink, could significantly improve cognitive function in people diagnosed with Mild Cognitive Impairment (MCI).
The results show that BrainXpert Energy Complex, a compound contained within BrainXpert, provides an alternative fuel source to glucose in the brain through the use of ketones and could reduce the symptoms experienced by people with MCI, such as forgetfulness and a decline in decision-making ability.
Despite affecting 15-20% of people aged 65+1 and being twice as prevalent as dementia2, MCI is still a largely unknown condition, with more than half (58%) of the UK having little or no knowledge of it3. BrainXpert is the first available treatment for people diagnosed with MCI.
Methodology
The new six-month randomised controlled BENEFIC (Brain ENErgy, Functional Imaging and Cognition) trial investigated the role of ketones, which can be used by the brain as an alternative fuel source to glucose, in the treatment of MCI.
In the randomised controlled BENEFIC trial, the intervention group was given the specialised ketogenic drink (BrainXpert Energy Complex) in a portion of 30g/day of ketogenic medium chain triglycerides (kMCT) that increased blood ketones. In comparison, the placebo group was provided a drink with an equivalent amount of energy that did not produce ketones – both drinks were taken twice a day for six months.
During the trial, episodic memory was evaluated using the French version of the 16-item free and cued word learning and recall test4 and the Brief Visual Memory Test-Revised (BVMT-R)5. For executive function, attention and processing speed, the Trail Making test, Stroop Colour and Word Interference test (Stroop), and the Verbal Fluency (VF) tests from the Delis-Kaplan Executive Function System were used respectively. In addition, the Boston Naming Test (TNT)6 was used for the assessment of language ability.
Trial results
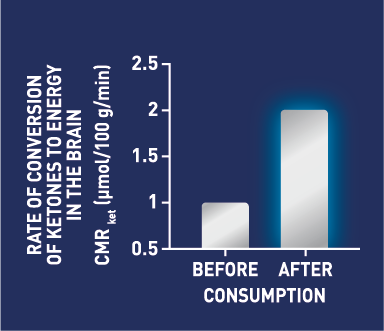
The trial results confirmed that an intervention with the BrainXpert specialised ketogenic drink clinically demonstrated a doubling of the ketones used by the brain, thereby significantly reducing the brain energy deficit caused by impaired brain glucose metabolism. Participants also showed a statistically significant reduction in MCI symptoms – they had improved memory, improved word recall, were able to think quicker, and were more able to multi-task versus participants that were given the placebo drink.
These ground-breaking results established for the first time that a specialised drink containing a compound rich in kMCT and milk protein is an effective alternative brain energy source for people living with MCI. This research signals a major breakthrough for the MCI community that, until now, has had no available treatments7.
Identifying significant improvement in cognitive function in patients with MCI is an exciting development that gives us great motivation to stay on this research track. This is only the beginning and the hope is that further ketogenic innovations can be found to not just boost brain function, but to slow down progression to Alzheimer’s disease and other forms of cognitive decline linked to ageing. These results should significantly improve the quality of life for people living with Mild Cognitive Impairment.
BENEFIC trial principal investigator Professor Stephen Cunnane, from the University of Sherbrooke, Canada
Phase one of the BENEFIC trial was published in Alzheimer’s & Dementia: The Journal of the Alzheimer’s Association in 2019, and the cognitive results of Phases 1 and 2 combined were published in the same journal in October 20208.
BrainXpert is now available online at http://www.brainxpert.com/. For more information on the study, or BrainXpert, please visit http://www.nestlehealthscience.com/MCI
BrainXpert is intended for special medical purposes for the dietary management of patients suffering from Mild Cognitive Impairment. To be used under medical supervision.
References
- Fortier, M., et al. A ketogenic drink improves brain energy and some measures of cognition in mild cognitive impairment Alzheimer’s & Dementia 2019;15: 625-634
https://doi.org/10.1016/j.jalz.2018.12.017 - World Health Organisation (2019). ‘Dementia’. Available at https://www.who.int/news-room/fact-sheets/detail/dementia
- Research conducted amongst 2,000 adults 18+ between 5th October and 8th October 2020.
- Van der Linden M, et al. L’épreuve de Rappel Libre/Rappel Indicé à 16 items (RL/RI-16). In: M. Van der Linden SA, & A. Agniel, editor. L’évaluation des troubles de la mémoire épisodique (avec leur étalonnage). Marseille (France): Solal; 2004. p. 25-47.
- Benedict R. Brief Visuospatial Memory Test-Revised professional manual. Odessa, FL: Psychological Assessment Resources; 1997.
- Kaplan E, Goodglass H, Weintraub S. The Boston Naming Test. Philadelphia: Lea & Febiger; 1983
- Fortier, M., et al. A ketogenic drink improves cognition in Mild Cognitive Impairment: results of a 6-month RCT. In press Alzheimer’s & Dementia 2020
https://doi.org/10.1002/alz.12206 - Fortier, M., et al. A ketogenic drink improves cognition in Mild Cognitive Impairment: results of a 6-month RCT. In press Alzheimer’s & Dementia 2020
https://doi.org/10.1002/alz.12206
